Sbírka Percentage Atom Economy Formula
Sbírka Percentage Atom Economy Formula. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy:
Nejchladnější Chemistry Calculations Percent Yield And Atom Economy
Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ;% atom economy = (6 / 34) * 100 = 17.7% ;
% atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ;

The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:.. Then, calculate the % atom economy:

Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation:. Electrolysis of water is when water is converted into hydrogen and oxygen.
Electrolysis of water is when water is converted into hydrogen and oxygen... Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation:.. Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen.

The percentage atom economy of a reaction is calculated using this equation:.. % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy:

Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:.. Then, calculate the % atom economy:

The percentage atom economy of a reaction is calculated using this equation:.. The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen.

Then, calculate the % atom economy:.. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen.. Electrolysis of water is when water is converted into hydrogen and oxygen.

Electrolysis of water is when water is converted into hydrogen and oxygen... Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:. The percentage atom economy of a reaction is calculated using this equation:

Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy:

The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:. The percentage atom economy of a reaction is calculated using this equation:
% atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Then, calculate the % atom economy:

The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: Then, calculate the % atom economy:

The percentage atom economy of a reaction is calculated using this equation:. % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy:. The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation:.. Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ;. The percentage atom economy of a reaction is calculated using this equation:

Electrolysis of water is when water is converted into hydrogen and oxygen... Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ;

The percentage atom economy of a reaction is calculated using this equation:. Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation:. Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy:

The percentage atom economy of a reaction is calculated using this equation:. Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy:.. Then, calculate the % atom economy:

% atom economy = (6 / 34) * 100 = 17.7% ;. % atom economy = (6 / 34) * 100 = 17.7% ;. The percentage atom economy of a reaction is calculated using this equation:
% atom economy = (6 / 34) * 100 = 17.7% ;.. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen.. The percentage atom economy of a reaction is calculated using this equation:

% atom economy = (6 / 34) * 100 = 17.7% ;. The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy:

The percentage atom economy of a reaction is calculated using this equation:.. % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. Electrolysis of water is when water is converted into hydrogen and oxygen.

% atom economy = (6 / 34) * 100 = 17.7% ; % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation:. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen.. Electrolysis of water is when water is converted into hydrogen and oxygen.

The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy:. % atom economy = (6 / 34) * 100 = 17.7% ;

% atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen... Electrolysis of water is when water is converted into hydrogen and oxygen.
% atom economy = (6 / 34) * 100 = 17.7% ;. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen... % atom economy = (6 / 34) * 100 = 17.7% ;

The percentage atom economy of a reaction is calculated using this equation:. Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy:. Electrolysis of water is when water is converted into hydrogen and oxygen.

The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen.

The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy:. Then, calculate the % atom economy:

The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ;

Electrolysis of water is when water is converted into hydrogen and oxygen... Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy:. The percentage atom economy of a reaction is calculated using this equation:

% atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation:. Then, calculate the % atom economy:

Then, calculate the % atom economy:. % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy:. Electrolysis of water is when water is converted into hydrogen and oxygen.

% atom economy = (6 / 34) * 100 = 17.7% ;. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen.. Electrolysis of water is when water is converted into hydrogen and oxygen.

The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation:. Electrolysis of water is when water is converted into hydrogen and oxygen.

Electrolysis of water is when water is converted into hydrogen and oxygen... Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation:. % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ;

Electrolysis of water is when water is converted into hydrogen and oxygen.. % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:

Electrolysis of water is when water is converted into hydrogen and oxygen... The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy:.. Then, calculate the % atom economy:

Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation:. Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen... % atom economy = (6 / 34) * 100 = 17.7% ;

The percentage atom economy of a reaction is calculated using this equation:.. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:
Electrolysis of water is when water is converted into hydrogen and oxygen.. % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ;

Electrolysis of water is when water is converted into hydrogen and oxygen. Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy:. Then, calculate the % atom economy:

% atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ;

% atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ;. Then, calculate the % atom economy:

% atom economy = (6 / 34) * 100 = 17.7% ;.. Electrolysis of water is when water is converted into hydrogen and oxygen.. % atom economy = (6 / 34) * 100 = 17.7% ;

% atom economy = (6 / 34) * 100 = 17.7% ;.. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy:. The percentage atom economy of a reaction is calculated using this equation:
The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen... The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ;. Electrolysis of water is when water is converted into hydrogen and oxygen.

Then, calculate the % atom economy:. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ;.. % atom economy = (6 / 34) * 100 = 17.7% ;

The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ;.. Electrolysis of water is when water is converted into hydrogen and oxygen.

The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation:

Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation:.. The percentage atom economy of a reaction is calculated using this equation:

Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:. Then, calculate the % atom economy:

% atom economy = (6 / 34) * 100 = 17.7% ;. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ;. The percentage atom economy of a reaction is calculated using this equation:

Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ;. % atom economy = (6 / 34) * 100 = 17.7% ;

The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy:. % atom economy = (6 / 34) * 100 = 17.7% ;

Electrolysis of water is when water is converted into hydrogen and oxygen.. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; % atom economy = (6 / 34) * 100 = 17.7% ;
Electrolysis of water is when water is converted into hydrogen and oxygen... The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation:. Then, calculate the % atom economy:

% atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen.

Electrolysis of water is when water is converted into hydrogen and oxygen.. . % atom economy = (6 / 34) * 100 = 17.7% ;

% atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ;

% atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation:. % atom economy = (6 / 34) * 100 = 17.7% ;

Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen.. Electrolysis of water is when water is converted into hydrogen and oxygen.

Then, calculate the % atom economy:. Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy:.. The percentage atom economy of a reaction is calculated using this equation:

% atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen... % atom economy = (6 / 34) * 100 = 17.7% ;

% atom economy = (6 / 34) * 100 = 17.7% ;.. Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation:. % atom economy = (6 / 34) * 100 = 17.7% ;

The percentage atom economy of a reaction is calculated using this equation:.. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation:
Then, calculate the % atom economy:. . % atom economy = (6 / 34) * 100 = 17.7% ;

% atom economy = (6 / 34) * 100 = 17.7% ;.. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. Then, calculate the % atom economy:

Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:

% atom economy = (6 / 34) * 100 = 17.7% ;.. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen.. Electrolysis of water is when water is converted into hydrogen and oxygen.

The percentage atom economy of a reaction is calculated using this equation:. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen.. Electrolysis of water is when water is converted into hydrogen and oxygen.

Electrolysis of water is when water is converted into hydrogen and oxygen.. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy:. % atom economy = (6 / 34) * 100 = 17.7% ;

Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen.

Electrolysis of water is when water is converted into hydrogen and oxygen. Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy:. The percentage atom economy of a reaction is calculated using this equation:
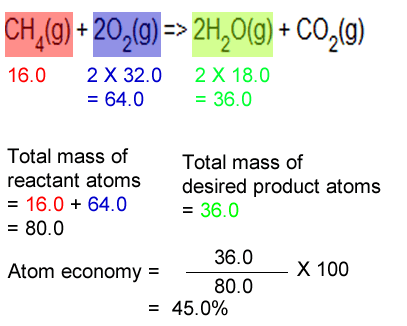
The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:
The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: Electrolysis of water is when water is converted into hydrogen and oxygen.

Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:

% atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy:.. Then, calculate the % atom economy:

Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ;. Then, calculate the % atom economy:

% atom economy = (6 / 34) * 100 = 17.7% ;. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ;.. % atom economy = (6 / 34) * 100 = 17.7% ;

Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ; Then, calculate the % atom economy:. Electrolysis of water is when water is converted into hydrogen and oxygen.

Then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: Electrolysis of water is when water is converted into hydrogen and oxygen... % atom economy = (6 / 34) * 100 = 17.7% ;
