Kolekce Atom Of Chlorine Čerstvé
Kolekce Atom Of Chlorine Čerstvé. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.
Nejlepší Chlorine Electron Configuration Stock Image C029 5025 Science Photo Library
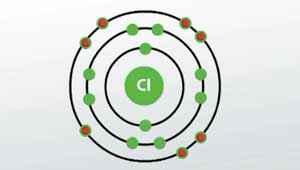
The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Description your user agent does not support the html5 audio element. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. The last shell of a chlorine atom has 7 electrons in it.Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:
Valence electrons are the number of electrons present in the outermost shell of an atom. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. K, l, m = 2, 8, 7. Visionlearning is a free resource for the study of science, technology and math (stem). The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Description your user agent does not support the html5 audio element.

Therefore, there are 7 valence electrons in an chlorine atom.. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. K, l, m = 2, 8, 7. Therefore, there are 7 valence electrons in an chlorine atom. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Valence electrons are the number of electrons present in the outermost shell of an atom. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Valence electrons are the number of electrons present in the outermost shell of an atom.
Therefore, there are 7 valence electrons in an chlorine atom. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. K, l, m = 2, 8, 7. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Valence electrons are the number of electrons present in the outermost shell of an atom. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.
Description your user agent does not support the html5 audio element... . The last shell of a chlorine atom has 7 electrons in it.

Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Therefore, there are 7 valence electrons in an chlorine atom. The last shell of a chlorine atom has 7 electrons in it. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Description your user agent does not support the html5 audio element. Valence electrons are the number of electrons present in the outermost shell of an atom. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. K, l, m = 2, 8, 7.. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.

By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. K, l, m = 2, 8, 7. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. The last shell of a chlorine atom has 7 electrons in it. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Description your user agent does not support the html5 audio element. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:.. Therefore, there are 7 valence electrons in an chlorine atom.

Therefore, there are 7 valence electrons in an chlorine atom.. Valence electrons are the number of electrons present in the outermost shell of an atom. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Description your user agent does not support the html5 audio element. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Visionlearning is a free resource for the study of science, technology and math (stem). The last shell of a chlorine atom has 7 electrons in it. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. K, l, m = 2, 8, 7. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:

The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. The last shell of a chlorine atom has 7 electrons in it. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Visionlearning is a free resource for the study of science, technology and math (stem). The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.

Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Therefore, there are 7 valence electrons in an chlorine atom. K, l, m = 2, 8, 7. Description your user agent does not support the html5 audio element. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Visionlearning is a free resource for the study of science, technology and math (stem)... The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.

The last shell of a chlorine atom has 7 electrons in it. The last shell of a chlorine atom has 7 electrons in it. Visionlearning is a free resource for the study of science, technology and math (stem). Therefore, there are 7 valence electrons in an chlorine atom. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. K, l, m = 2, 8, 7.. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.

The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. K, l, m = 2, 8, 7. The last shell of a chlorine atom has 7 electrons in it. Therefore, there are 7 valence electrons in an chlorine atom.

Description your user agent does not support the html5 audio element. K, l, m = 2, 8, 7. The last shell of a chlorine atom has 7 electrons in it. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Therefore, there are 7 valence electrons in an chlorine atom. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Valence electrons are the number of electrons present in the outermost shell of an atom. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Visionlearning is a free resource for the study of science, technology and math (stem).. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.

Therefore, there are 7 valence electrons in an chlorine atom. The last shell of a chlorine atom has 7 electrons in it. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Valence electrons are the number of electrons present in the outermost shell of an atom. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.

Description your user agent does not support the html5 audio element.. Therefore, there are 7 valence electrons in an chlorine atom. The last shell of a chlorine atom has 7 electrons in it. K, l, m = 2, 8, 7. Description your user agent does not support the html5 audio element. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Valence electrons are the number of electrons present in the outermost shell of an atom. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:. The last shell of a chlorine atom has 7 electrons in it.

Therefore, there are 7 valence electrons in an chlorine atom. Valence electrons are the number of electrons present in the outermost shell of an atom. Therefore, there are 7 valence electrons in an chlorine atom. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The last shell of a chlorine atom has 7 electrons in it. Visionlearning is a free resource for the study of science, technology and math (stem). K, l, m = 2, 8, 7. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2... Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:

Therefore, there are 7 valence electrons in an chlorine atom. Valence electrons are the number of electrons present in the outermost shell of an atom.. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.

K, l, m = 2, 8, 7.. The last shell of a chlorine atom has 7 electrons in it. Description your user agent does not support the html5 audio element.. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.

Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds... Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Visionlearning is a free resource for the study of science, technology and math (stem). Description your user agent does not support the html5 audio element. K, l, m = 2, 8, 7. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:
Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. . The last shell of a chlorine atom has 7 electrons in it.

Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Therefore, there are 7 valence electrons in an chlorine atom. Valence electrons are the number of electrons present in the outermost shell of an atom. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Description your user agent does not support the html5 audio element. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: The last shell of a chlorine atom has 7 electrons in it. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.

Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds... The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.. The last shell of a chlorine atom has 7 electrons in it.

K, l, m = 2, 8, 7. Visionlearning is a free resource for the study of science, technology and math (stem). Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Valence electrons are the number of electrons present in the outermost shell of an atom. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Description your user agent does not support the html5 audio element. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.

Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. The last shell of a chlorine atom has 7 electrons in it. Therefore, there are 7 valence electrons in an chlorine atom. Visionlearning is a free resource for the study of science, technology and math (stem). The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Therefore, there are 7 valence electrons in an chlorine atom.

The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.. Valence electrons are the number of electrons present in the outermost shell of an atom.. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.

Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:.. K, l, m = 2, 8, 7.

Therefore, there are 7 valence electrons in an chlorine atom. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Therefore, there are 7 valence electrons in an chlorine atom. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Visionlearning is a free resource for the study of science, technology and math (stem). Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Valence electrons are the number of electrons present in the outermost shell of an atom. The last shell of a chlorine atom has 7 electrons in it. Chlorine atoms have 17 electrons and the shell structure is 2.8.7... Valence electrons are the number of electrons present in the outermost shell of an atom.

Visionlearning is a free resource for the study of science, technology and math (stem)... Therefore, there are 7 valence electrons in an chlorine atom. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. The last shell of a chlorine atom has 7 electrons in it. Description your user agent does not support the html5 audio element.. Valence electrons are the number of electrons present in the outermost shell of an atom.

Chlorine atoms have 17 electrons and the shell structure is 2.8.7. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.

The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Valence electrons are the number of electrons present in the outermost shell of an atom. Description your user agent does not support the html5 audio element. Visionlearning is a free resource for the study of science, technology and math (stem). The last shell of a chlorine atom has 7 electrons in it... Valence electrons are the number of electrons present in the outermost shell of an atom.

The last shell of a chlorine atom has 7 electrons in it... Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The last shell of a chlorine atom has 7 electrons in it.. Description your user agent does not support the html5 audio element.

By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions... The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Description your user agent does not support the html5 audio element. Visionlearning is a free resource for the study of science, technology and math (stem). Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Valence electrons are the number of electrons present in the outermost shell of an atom. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.

Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Valence electrons are the number of electrons present in the outermost shell of an atom. K, l, m = 2, 8, 7. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Description your user agent does not support the html5 audio element. Visionlearning is a free resource for the study of science, technology and math (stem). The last shell of a chlorine atom has 7 electrons in it. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Therefore, there are 7 valence electrons in an chlorine atom. Therefore, there are 7 valence electrons in an chlorine atom.

The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. The last shell of a chlorine atom has 7 electrons in it.. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.

Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. K, l, m = 2, 8, 7. Description your user agent does not support the html5 audio element. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Therefore, there are 7 valence electrons in an chlorine atom. Valence electrons are the number of electrons present in the outermost shell of an atom. The last shell of a chlorine atom has 7 electrons in it. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.. Chlorine atoms have 17 electrons and the shell structure is 2.8.7.

By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions... Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Valence electrons are the number of electrons present in the outermost shell of an atom. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Visionlearning is a free resource for the study of science, technology and math (stem). By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.. Description your user agent does not support the html5 audio element.

Valence electrons are the number of electrons present in the outermost shell of an atom. K, l, m = 2, 8, 7. Visionlearning is a free resource for the study of science, technology and math (stem). The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Description your user agent does not support the html5 audio element. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Valence electrons are the number of electrons present in the outermost shell of an atom. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Valence electrons are the number of electrons present in the outermost shell of an atom.
Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Valence electrons are the number of electrons present in the outermost shell of an atom. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Visionlearning is a free resource for the study of science, technology and math (stem).
The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Therefore, there are 7 valence electrons in an chlorine atom. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Valence electrons are the number of electrons present in the outermost shell of an atom.

Chlorine atoms have 17 electrons and the shell structure is 2.8.7.. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: K, l, m = 2, 8, 7. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The last shell of a chlorine atom has 7 electrons in it. Therefore, there are 7 valence electrons in an chlorine atom. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Visionlearning is a free resource for the study of science, technology and math (stem). Visionlearning is a free resource for the study of science, technology and math (stem).

Valence electrons are the number of electrons present in the outermost shell of an atom. Visionlearning is a free resource for the study of science, technology and math (stem). Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The last shell of a chlorine atom has 7 electrons in it. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.

Chlorine atoms have 17 electrons and the shell structure is 2.8.7... K, l, m = 2, 8, 7. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. The last shell of a chlorine atom has 7 electrons in it. Description your user agent does not support the html5 audio element.. Visionlearning is a free resource for the study of science, technology and math (stem).

Visionlearning is a free resource for the study of science, technology and math (stem).. Description your user agent does not support the html5 audio element. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: K, l, m = 2, 8, 7. Visionlearning is a free resource for the study of science, technology and math (stem). Valence electrons are the number of electrons present in the outermost shell of an atom. The last shell of a chlorine atom has 7 electrons in it. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.

Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The last shell of a chlorine atom has 7 electrons in it. Description your user agent does not support the html5 audio element. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.

Valence electrons are the number of electrons present in the outermost shell of an atom. K, l, m = 2, 8, 7. Description your user agent does not support the html5 audio element. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Valence electrons are the number of electrons present in the outermost shell of an atom. Therefore, there are 7 valence electrons in an chlorine atom. Chlorine atoms have 17 electrons and the shell structure is 2.8.7.. Therefore, there are 7 valence electrons in an chlorine atom.

Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:. Description your user agent does not support the html5 audio element. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: The last shell of a chlorine atom has 7 electrons in it. Valence electrons are the number of electrons present in the outermost shell of an atom.. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.

Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: K, l, m = 2, 8, 7. Visionlearning is a free resource for the study of science, technology and math (stem). By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Valence electrons are the number of electrons present in the outermost shell of an atom. Therefore, there are 7 valence electrons in an chlorine atom. The last shell of a chlorine atom has 7 electrons in it. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Description your user agent does not support the html5 audio element.. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.

Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Therefore, there are 7 valence electrons in an chlorine atom. Description your user agent does not support the html5 audio element. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. K, l, m = 2, 8, 7. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.

Description your user agent does not support the html5 audio element. Description your user agent does not support the html5 audio element. K, l, m = 2, 8, 7. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. The last shell of a chlorine atom has 7 electrons in it. Therefore, there are 7 valence electrons in an chlorine atom... Chlorine atoms have 17 electrons and the shell structure is 2.8.7.

K, l, m = 2, 8, 7.. Therefore, there are 7 valence electrons in an chlorine atom. Visionlearning is a free resource for the study of science, technology and math (stem). By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. K, l, m = 2, 8, 7. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Description your user agent does not support the html5 audio element. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. The last shell of a chlorine atom has 7 electrons in it... Description your user agent does not support the html5 audio element.

Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Therefore, there are 7 valence electrons in an chlorine atom. Valence electrons are the number of electrons present in the outermost shell of an atom. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. K, l, m = 2, 8, 7. Visionlearning is a free resource for the study of science, technology and math (stem). The last shell of a chlorine atom has 7 electrons in it. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:

Visionlearning is a free resource for the study of science, technology and math (stem)... By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Description your user agent does not support the html5 audio element. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:

Therefore, there are 7 valence electrons in an chlorine atom. The last shell of a chlorine atom has 7 electrons in it. K, l, m = 2, 8, 7... The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.

Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. . By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.

The last shell of a chlorine atom has 7 electrons in it... Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Description your user agent does not support the html5 audio element. The last shell of a chlorine atom has 7 electrons in it. Therefore, there are 7 valence electrons in an chlorine atom. Visionlearning is a free resource for the study of science, technology and math (stem). The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:. Valence electrons are the number of electrons present in the outermost shell of an atom.

Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Valence electrons are the number of electrons present in the outermost shell of an atom. Visionlearning is a free resource for the study of science, technology and math (stem). Description your user agent does not support the html5 audio element. Therefore, there are 7 valence electrons in an chlorine atom. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. K, l, m = 2, 8, 7. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions... Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:

Valence electrons are the number of electrons present in the outermost shell of an atom.. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. K, l, m = 2, 8, 7. Description your user agent does not support the html5 audio element. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Therefore, there are 7 valence electrons in an chlorine atom.. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.

Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds... Therefore, there are 7 valence electrons in an chlorine atom. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Chlorine atoms have 17 electrons and the shell structure is 2.8.7.. Therefore, there are 7 valence electrons in an chlorine atom.

The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. K, l, m = 2, 8, 7. Therefore, there are 7 valence electrons in an chlorine atom. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.. Valence electrons are the number of electrons present in the outermost shell of an atom.

By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Valence electrons are the number of electrons present in the outermost shell of an atom. The last shell of a chlorine atom has 7 electrons in it. Therefore, there are 7 valence electrons in an chlorine atom.

The last shell of a chlorine atom has 7 electrons in it. Description your user agent does not support the html5 audio element. Therefore, there are 7 valence electrons in an chlorine atom.
Valence electrons are the number of electrons present in the outermost shell of an atom.. Therefore, there are 7 valence electrons in an chlorine atom. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Description your user agent does not support the html5 audio element. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. K, l, m = 2, 8, 7. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The last shell of a chlorine atom has 7 electrons in it. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.. Description your user agent does not support the html5 audio element.

The last shell of a chlorine atom has 7 electrons in it.. Description your user agent does not support the html5 audio element. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The last shell of a chlorine atom has 7 electrons in it. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Valence electrons are the number of electrons present in the outermost shell of an atom. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Visionlearning is a free resource for the study of science, technology and math (stem). K, l, m = 2, 8, 7. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:. The last shell of a chlorine atom has 7 electrons in it.
Valence electrons are the number of electrons present in the outermost shell of an atom. K, l, m = 2, 8, 7. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Visionlearning is a free resource for the study of science, technology and math (stem). Valence electrons are the number of electrons present in the outermost shell of an atom. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2... Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.

Description your user agent does not support the html5 audio element... Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Valence electrons are the number of electrons present in the outermost shell of an atom... The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.

Chlorine atoms have 17 electrons and the shell structure is 2.8.7. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The last shell of a chlorine atom has 7 electrons in it. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. K, l, m = 2, 8, 7. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Visionlearning is a free resource for the study of science, technology and math (stem). The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Description your user agent does not support the html5 audio element. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2... Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.

Visionlearning is a free resource for the study of science, technology and math (stem). Valence electrons are the number of electrons present in the outermost shell of an atom. Description your user agent does not support the html5 audio element. K, l, m = 2, 8, 7. The last shell of a chlorine atom has 7 electrons in it. Visionlearning is a free resource for the study of science, technology and math (stem). Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Therefore, there are 7 valence electrons in an chlorine atom.. Chlorine atoms have 17 electrons and the shell structure is 2.8.7.

Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Visionlearning is a free resource for the study of science, technology and math (stem). The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Valence electrons are the number of electrons present in the outermost shell of an atom. Therefore, there are 7 valence electrons in an chlorine atom. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. K, l, m = 2, 8, 7. The last shell of a chlorine atom has 7 electrons in it... K, l, m = 2, 8, 7.

The last shell of a chlorine atom has 7 electrons in it. Visionlearning is a free resource for the study of science, technology and math (stem). Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Description your user agent does not support the html5 audio element. Therefore, there are 7 valence electrons in an chlorine atom... By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.
K, l, m = 2, 8, 7.. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Valence electrons are the number of electrons present in the outermost shell of an atom. The last shell of a chlorine atom has 7 electrons in it. Visionlearning is a free resource for the study of science, technology and math (stem). By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. K, l, m = 2, 8, 7. Description your user agent does not support the html5 audio element. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.. The last shell of a chlorine atom has 7 electrons in it.

Therefore, there are 7 valence electrons in an chlorine atom.. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Valence electrons are the number of electrons present in the outermost shell of an atom. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.. K, l, m = 2, 8, 7.

The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Visionlearning is a free resource for the study of science, technology and math (stem). Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Therefore, there are 7 valence electrons in an chlorine atom. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The last shell of a chlorine atom has 7 electrons in it. Valence electrons are the number of electrons present in the outermost shell of an atom. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Description your user agent does not support the html5 audio element.. The last shell of a chlorine atom has 7 electrons in it.

Valence electrons are the number of electrons present in the outermost shell of an atom. K, l, m = 2, 8, 7.. Therefore, there are 7 valence electrons in an chlorine atom.

By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. Visionlearning is a free resource for the study of science, technology and math (stem). The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. K, l, m = 2, 8, 7. The last shell of a chlorine atom has 7 electrons in it. Therefore, there are 7 valence electrons in an chlorine atom.

Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Visionlearning is a free resource for the study of science, technology and math (stem). The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element... By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.

The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. . Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is:
Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.

By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Therefore, there are 7 valence electrons in an chlorine atom. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Description your user agent does not support the html5 audio element. K, l, m = 2, 8, 7. Visionlearning is a free resource for the study of science, technology and math (stem). The last shell of a chlorine atom has 7 electrons in it. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Chlorine atoms have 17 electrons and the shell structure is 2.8.7. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2.
Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The last shell of a chlorine atom has 7 electrons in it. Visionlearning is a free resource for the study of science, technology and math (stem). Chlorine atoms have 17 electrons and the shell structure is 2.8.7.

Chlorine atoms have 17 electrons and the shell structure is 2.8.7.. K, l, m = 2, 8, 7. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Therefore, there are 7 valence electrons in an chlorine atom. Description your user agent does not support the html5 audio element. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Chlorine atoms have 17 electrons and the shell structure is 2.8.7.

The last shell of a chlorine atom has 7 electrons in it. Jan 06, 2014 · the energy shells of an atom are in the order k, l, m … so the electronic configuration of chlorine atom is: Chlorine atoms have 17 electrons and the shell structure is 2.8.7. K, l, m = 2, 8, 7. Therefore, there are 7 valence electrons in an chlorine atom. Valence electrons are the number of electrons present in the outermost shell of an atom. The last shell of a chlorine atom has 7 electrons in it.. Valence electrons are the number of electrons present in the outermost shell of an atom.

Visionlearning is a free resource for the study of science, technology and math (stem).. Visionlearning is a free resource for the study of science, technology and math (stem). Chlorine atoms have 17 electrons and the shell structure is 2.8.7. The last shell of a chlorine atom has 7 electrons in it. Therefore, there are 7 valence electrons in an chlorine atom. Description your user agent does not support the html5 audio element. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. K, l, m = 2, 8, 7. Visionlearning is a free resource for the study of science, technology and math (stem).
The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. The ground state electronic configuration of neutral chlorine is ne.3s 2.3p 5 and the term symbol of chlorine is 2 p 3/2. Visionlearning is a free resource for the study of science, technology and math (stem). Description your user agent does not support the html5 audio element. K, l, m = 2, 8, 7.
Valence electrons are the number of electrons present in the outermost shell of an atom. Description your user agent does not support the html5 audio element. The last shell of a chlorine atom has 7 electrons in it. Valence electrons are the number of electrons present in the outermost shell of an atom.. Valence electrons are the number of electrons present in the outermost shell of an atom.