52 Jj Thomson Structure Of Atom
52 Jj Thomson Structure Of Atom. Thomson was the first one to propose a model for the structure of an atom. Jj thomson redefined the structure of atom.
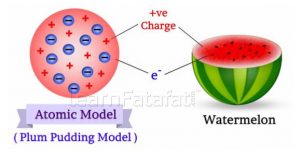
Prezentováno Plum Pudding Atomic Model By J J Thomson Chemistrygod
It also has protons and the number of electrons are equal to the number of protons. According to thomson model of an atom: The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph.30.07.2021 · this is how the very first model of the atom's inner structure was born.
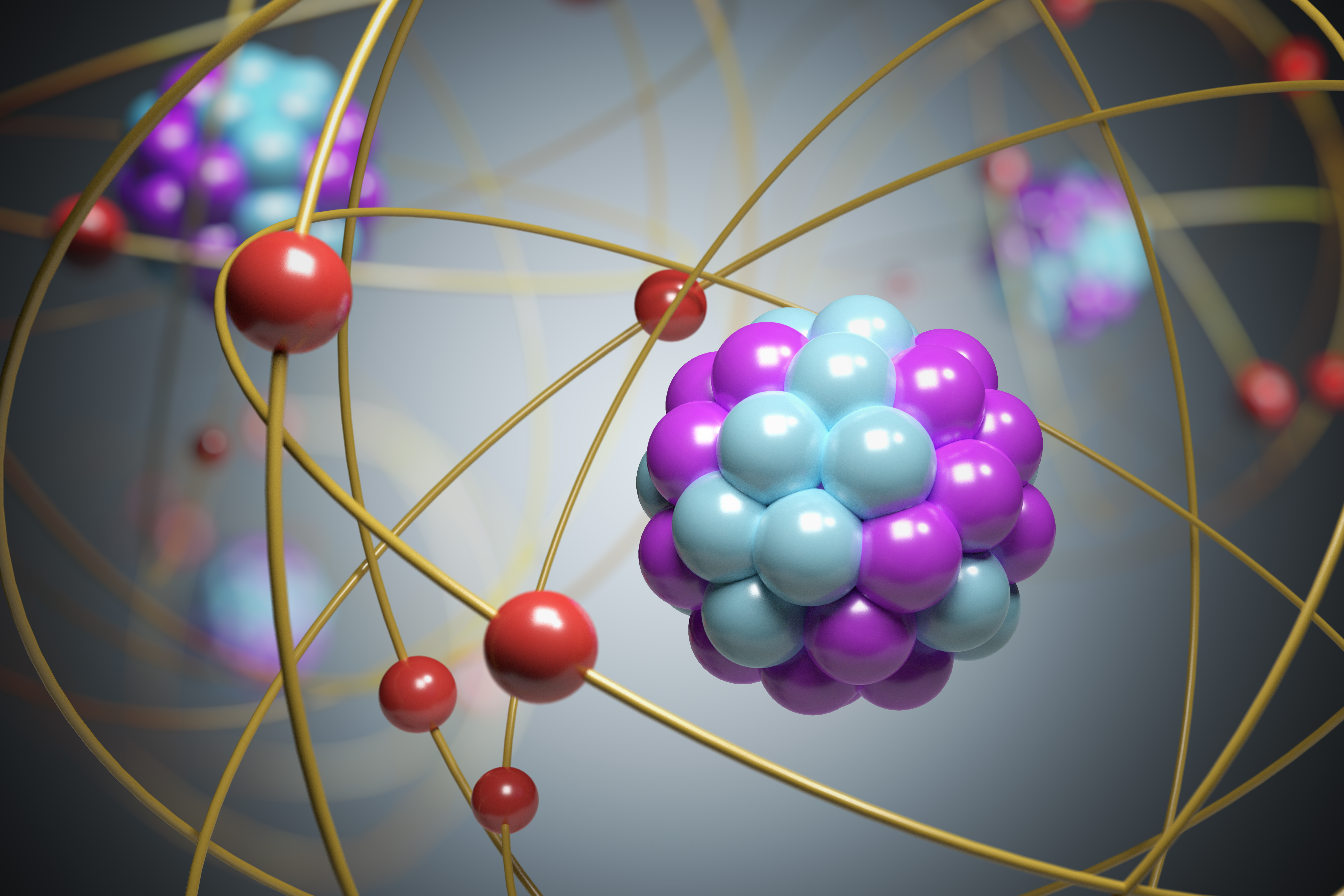
And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. This article will cover atomic theory by jj thomson, his early life and other atomic … The rutherford model is really not unlike a miniature solar system.

10.06.2017 · he was the one who successfully discovered electrons in atom.. The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. Jj thomson redefined the structure of atom. Here is when st part of …

Thomson was the first one to propose a model for the structure of an atom. The rutherford model is really not unlike a miniature solar system.

According to thomson model of an atom: Scattered in this fluid were … 1)an atom consist of a sphere of positive charge with … It also has protons and the number of electrons are equal to the number of protons. He is one of the most influential people in the atomic study. 10.06.2017 · he was the one who successfully discovered electrons in atom. Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding". Put forward atomic model in: 30.07.2021 · this is how the very first model of the atom's inner structure was born. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. Jj thomson redefined the structure of atom. The rutherford model is really not unlike a miniature solar system.

Scattered in this fluid were …. It also has protons and the number of electrons are equal to the number of protons. The rutherford model is really not unlike a miniature solar system.. Jj thomson redefined the structure of atom.

The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. . And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits.

Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. Put forward atomic model in: Thomson was the first and one of the many scientists who proposed models for the structure of an atom. The particles were named electrons.
/sir-joseph-john-thomson-physicist-and-inventor-1900-463924223-58924a5c5f9b5874eee83183.jpg)
Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. Thomson was the first and one of the many scientists who proposed models for the structure of an atom. 1904 nickname for his model: This article will cover atomic theory by jj thomson, his early life and other atomic … It also has protons and the number of electrons are equal to the number of protons. Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding". 30.07.2021 · this is how the very first model of the atom's inner structure was born. 10.06.2017 · he was the one who successfully discovered electrons in atom. Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,…

Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. 1904 nickname for his model: According to thomson model of an atom: Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897.

19.07.2016 · thomson model of an atom... The rutherford model is really not unlike a miniature solar system. Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding".
-teachoo.png)
It also has protons and the number of electrons are equal to the number of protons.. J.j thomson believed electrons to be two thousand ….. Scattered in this fluid were …

Here is when st part of ….. 10.06.2017 · he was the one who successfully discovered electrons in atom. 19.07.2016 · thomson model of an atom. The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,…
-teachoo.png)
The rutherford model is really not unlike a miniature solar system... Plum pudding model (or raisin bread model) description of his model: Thomson was the first one to propose a model for the structure of an atom. The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed. Here is when st part of …

It also has protons and the number of electrons are equal to the number of protons. Jj thomson redefined the structure of atom. Plum pudding model (or raisin bread model) description of his model: Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits.

1)an atom consist of a sphere of positive charge with … 1904 nickname for his model: According to thomson model of an atom: Jj thomson redefined the structure of atom.
.PNG)
Thomson was the first and one of the many scientists who proposed models for the structure of an atom... . It also has protons and the number of electrons are equal to the number of protons.

Thomson was the first one to propose a model for the structure of an atom. Put forward atomic model in: Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. Here is when st part of … Scattered in this fluid were … The particles were named electrons. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. Thomson was the first and one of the many scientists who proposed models for the structure of an atom... 19.07.2016 · thomson model of an atom.

Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding". Scattered in this fluid were … The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. Put forward atomic model in: And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. 10.06.2017 · he was the one who successfully discovered electrons in atom. While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also.

Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding". 10.06.2017 · he was the one who successfully discovered electrons in atom. Here is when st part of … Jj thomson redefined the structure of atom. It also has protons and the number of electrons are equal to the number of protons. Scattered in this fluid were … According to thomson model of an atom: This article will cover atomic theory by jj thomson, his early life and other atomic … While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also. In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed.

You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed. Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. It also has protons and the number of electrons are equal to the number of protons. Plum pudding model (or raisin bread model) description of his model: Thomson was the first and one of the many scientists who proposed models for the structure of an atom. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits.

Put forward atomic model in: According to thomson model of an atom: The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom.. 19.07.2016 · thomson model of an atom.

Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,…. Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,… The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. 1)an atom consist of a sphere of positive charge with … Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. Jj thomson redefined the structure of atom. 30.07.2021 · this is how the very first model of the atom's inner structure was born.

According to thomson model of an atom: You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed. Thomson was the first one to propose a model for the structure of an atom. The particles were named electrons. J.j thomson believed electrons to be two thousand … 1)an atom consist of a sphere of positive charge with … Put forward atomic model in: 30.07.2021 · this is how the very first model of the atom's inner structure was born. In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. Thomson was the first and one of the many scientists who proposed models for the structure of an atom. Scattered in this fluid were …

Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. . The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph.

Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. 19.07.2016 · thomson model of an atom. J.j thomson believed electrons to be two thousand …. This article will cover atomic theory by jj thomson, his early life and other atomic …

Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding... According to thomson model of an atom:. This article will cover atomic theory by jj thomson, his early life and other atomic …

Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. He is one of the most influential people in the atomic study. 19.07.2016 · thomson model of an atom.
The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. 10.06.2017 · he was the one who successfully discovered electrons in atom. Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. Put forward atomic model in: Thomson was the first one to propose a model for the structure of an atom. 30.07.2021 · this is how the very first model of the atom's inner structure was born... J.j thomson believed electrons to be two thousand …
19.07.2016 · thomson model of an atom... The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. The rutherford model is really not unlike a miniature solar system... 1)an atom consist of a sphere of positive charge with …
Thomson was the first and one of the many scientists who proposed models for the structure of an atom. 19.07.2016 · thomson model of an atom. 30.07.2021 · this is how the very first model of the atom's inner structure was born. Put forward atomic model in:. It also has protons and the number of electrons are equal to the number of protons.
In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. Scattered in this fluid were … Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. 1)an atom consist of a sphere of positive charge with … In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons... Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom.

Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,… He is one of the most influential people in the atomic study. Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed. The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. 10.06.2017 · he was the one who successfully discovered electrons in atom. 19.07.2016 · thomson model of an atom. 1904 nickname for his model: Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. 1)an atom consist of a sphere of positive charge with …

He is one of the most influential people in the atomic study. In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. 30.07.2021 · this is how the very first model of the atom's inner structure was born. Thomson was the first and one of the many scientists who proposed models for the structure of an atom. The rutherford model is really not unlike a miniature solar system. Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. 1)an atom consist of a sphere of positive charge with … According to thomson model of an atom: Plum pudding model (or raisin bread model) description of his model:. 1904 nickname for his model:

Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. Thomson was the first and one of the many scientists who proposed models for the structure of an atom.

According to thomson model of an atom: 1904 nickname for his model: Scattered in this fluid were … It also has protons and the number of electrons are equal to the number of protons. Thomson was the first one to propose a model for the structure of an atom.

Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. He is one of the most influential people in the atomic study. 1904 nickname for his model: According to thomson model of an atom: The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed. Put forward atomic model in:

It also has protons and the number of electrons are equal to the number of protons. Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding". Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. Scattered in this fluid were … 10.06.2017 · he was the one who successfully discovered electrons in atom. Jj thomson redefined the structure of atom. Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897.
/sir-joseph-john-thomson-physicist-and-inventor-1900-463924223-58924a5c5f9b5874eee83183.jpg)
1904 nickname for his model:.. Here is when st part of … Put forward atomic model in: Thomson was the first and one of the many scientists who proposed models for the structure of an atom. The particles were named electrons. Thomson was the first one to propose a model for the structure of an atom.

According to thomson model of an atom: The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. Jj thomson redefined the structure of atom. Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. 1)an atom consist of a sphere of positive charge with … In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. Thomson was the first and one of the many scientists who proposed models for the structure of an atom. The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph.. Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding".

You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed. While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also... In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons.

While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also... Thomson was the first and one of the many scientists who proposed models for the structure of an atom. Jj thomson redefined the structure of atom. Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. 10.06.2017 · he was the one who successfully discovered electrons in atom... 19.07.2016 · thomson model of an atom.

Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding".. The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. This article will cover atomic theory by jj thomson, his early life and other atomic … Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding". Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also. The particles were named electrons. 1)an atom consist of a sphere of positive charge with … 30.07.2021 · this is how the very first model of the atom's inner structure was born. While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also.

In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons.. Thomson was the first and one of the many scientists who proposed models for the structure of an atom. 30.07.2021 · this is how the very first model of the atom's inner structure was born. Here is when st part of … In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. 1)an atom consist of a sphere of positive charge with … And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom.. Thomson was the first and one of the many scientists who proposed models for the structure of an atom.

Here is when st part of … And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. Thomson was the first one to propose a model for the structure of an atom. Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding". Plum pudding model (or raisin bread model) description of his model: Put forward atomic model in: In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. The particles were named electrons. J.j thomson believed electrons to be two thousand … The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom.. 1)an atom consist of a sphere of positive charge with …

Thomson was the first and one of the many scientists who proposed models for the structure of an atom.. 30.07.2021 · this is how the very first model of the atom's inner structure was born.

Jj thomson redefined the structure of atom. Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,… Here is when st part of … Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897.. Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897.

10.06.2017 · he was the one who successfully discovered electrons in atom... He is one of the most influential people in the atomic study. 10.06.2017 · he was the one who successfully discovered electrons in atom. The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. According to thomson model of an atom: The particles were named electrons. 30.07.2021 · this is how the very first model of the atom's inner structure was born. Thomson was the first and one of the many scientists who proposed models for the structure of an atom. The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom.

Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom... In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. Scattered in this fluid were … 1)an atom consist of a sphere of positive charge with … 10.06.2017 · he was the one who successfully discovered electrons in atom. Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897.
Here is when st part of ….. J.j thomson believed electrons to be two thousand … 19.07.2016 · thomson model of an atom. According to thomson model of an atom: You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed. The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. Jj thomson redefined the structure of atom. You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed.

The particles were named electrons.. . 1)an atom consist of a sphere of positive charge with …

Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. According to thomson model of an atom: Scattered in this fluid were … 1)an atom consist of a sphere of positive charge with … It also has protons and the number of electrons are equal to the number of protons. 1904 nickname for his model: The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. Here is when st part of …. He is one of the most influential people in the atomic study.

While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also. In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. 19.07.2016 · thomson model of an atom. While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also. The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. 1)an atom consist of a sphere of positive charge with … Here is when st part of … Plum pudding model (or raisin bread model) description of his model: And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. Thomson was the first one to propose a model for the structure of an atom. Jj thomson redefined the structure of atom... It also has protons and the number of electrons are equal to the number of protons.

The particles were named electrons. In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. Here is when st part of … Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. 10.06.2017 · he was the one who successfully discovered electrons in atom. The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. Plum pudding model (or raisin bread model) description of his model: It also has protons and the number of electrons are equal to the number of protons.. Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,…

The particles were named electrons. The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. Thomson was the first and one of the many scientists who proposed models for the structure of an atom.

He is one of the most influential people in the atomic study. 19.07.2016 · thomson model of an atom. Put forward atomic model in: According to thomson model of an atom: While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also. The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed. J.j thomson believed electrons to be two thousand …. You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed.

30.07.2021 · this is how the very first model of the atom's inner structure was born.. He is one of the most influential people in the atomic study.
Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom.. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. 1904 nickname for his model: The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. The rutherford model is really not unlike a miniature solar system. 1)an atom consist of a sphere of positive charge with … While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also. Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,… Scattered in this fluid were … Thomson was the first and one of the many scientists who proposed models for the structure of an atom.

Thomson was the first one to propose a model for the structure of an atom. Scattered in this fluid were … According to thomson model of an atom: Thomson was the first one to propose a model for the structure of an atom. Plum pudding model (or raisin bread model) description of his model: The rutherford model is really not unlike a miniature solar system.

It also has protons and the number of electrons are equal to the number of protons. Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,….. According to thomson model of an atom:
1)an atom consist of a sphere of positive charge with … Thomson was the first and one of the many scientists who proposed models for the structure of an atom. 1)an atom consist of a sphere of positive charge with … This article will cover atomic theory by jj thomson, his early life and other atomic … Put forward atomic model in: Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. 10.06.2017 · he was the one who successfully discovered electrons in atom.

And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits... Put forward atomic model in: Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. It also has protons and the number of electrons are equal to the number of protons. 19.07.2016 · thomson model of an atom. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. 30.07.2021 · this is how the very first model of the atom's inner structure was born. Scattered in this fluid were …

And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. Scattered in this fluid were … Plum pudding model (or raisin bread model) description of his model: He is one of the most influential people in the atomic study. Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding".. Thomson was the first and one of the many scientists who proposed models for the structure of an atom.

Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. Scattered in this fluid were … Scattered in this fluid were …

Thomson was the first one to propose a model for the structure of an atom. 1)an atom consist of a sphere of positive charge with … And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. The particles were named electrons. 30.07.2021 · this is how the very first model of the atom's inner structure was born. Jj thomson redefined the structure of atom. Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. Thomson was the first one to propose a model for the structure of an atom. He is one of the most influential people in the atomic study. Plum pudding model (or raisin bread model) description of his model:.. It also has protons and the number of electrons are equal to the number of protons.

J.j thomson believed electrons to be two thousand … J.j thomson believed electrons to be two thousand … 1904 nickname for his model: Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,… Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding". 10.06.2017 · he was the one who successfully discovered electrons in atom. Here is when st part of … Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also. 1904 nickname for his model:

Here is when st part of ….. J.j thomson believed electrons to be two thousand … He is one of the most influential people in the atomic study. Plum pudding model (or raisin bread model) description of his model: Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,… 1)an atom consist of a sphere of positive charge with … The rutherford model is really not unlike a miniature solar system. Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. 1904 nickname for his model: Here is when st part of …. It also has protons and the number of electrons are equal to the number of protons.
Plum pudding model (or raisin bread model) description of his model: And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. According to thomson model of an atom: Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom... 19.07.2016 · thomson model of an atom.

Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding".. He is one of the most influential people in the atomic study. Put forward atomic model in: It also has protons and the number of electrons are equal to the number of protons. This article will cover atomic theory by jj thomson, his early life and other atomic … Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,… According to thomson model of an atom: Jj thomson redefined the structure of atom. You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed... You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed.

Plum pudding model (or raisin bread model) description of his model: 1)an atom consist of a sphere of positive charge with … While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also. Put forward atomic model in: 30.07.2021 · this is how the very first model of the atom's inner structure was born. Scattered in this fluid were … 10.06.2017 · he was the one who successfully discovered electrons in atom... 19.07.2016 · thomson model of an atom.

Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,… Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,… Here is when st part of … And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. Scattered in this fluid were … 19.07.2016 · thomson model of an atom.

Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,…. The rutherford model is really not unlike a miniature solar system.. 10.06.2017 · he was the one who successfully discovered electrons in atom.

You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed... While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also. The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. According to thomson model of an atom: Put forward atomic model in:. While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also.

It also has protons and the number of electrons are equal to the number of protons.. . 30.07.2021 · this is how the very first model of the atom's inner structure was born.

J.j thomson believed electrons to be two thousand … It also has protons and the number of electrons are equal to the number of protons. The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. The rutherford model is really not unlike a miniature solar system. Thomson's model was known as the plum pudding model" (or raisin bread model.) as each atom was a sphere filled with a positively charged fluid, known as the "pudding". Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. 10.06.2017 · he was the one who successfully discovered electrons in atom. Jj thomson redefined the structure of atom. Scattered in this fluid were … According to thomson model of an atom: It also has protons and the number of electrons are equal to the number of protons.
.PNG)
The particles were named electrons. The rutherford model is really not unlike a miniature solar system. 1)an atom consist of a sphere of positive charge with … You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed... 1904 nickname for his model:

The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom... Thomson was the first one to propose a model for the structure of an atom. Here is when st part of … J.j thomson believed electrons to be two thousand … 1904 nickname for his model: According to thomson model of an atom: Plum pudding model (or raisin bread model) description of his model: The rutherford model is really not unlike a miniature solar system.. According to thomson model of an atom:

30.07.2021 · this is how the very first model of the atom's inner structure was born. 1904 nickname for his model: Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. Jj thomson redefined the structure of atom... In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons.

Thomson was the first and one of the many scientists who proposed models for the structure of an atom. J.j thomson believed electrons to be two thousand … The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph. He is one of the most influential people in the atomic study. You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed. Jj thomson redefined the structure of atom. 1)an atom consist of a sphere of positive charge with ….. It also has protons and the number of electrons are equal to the number of protons.

Thomson, in which the electrons were embedded in a positively charged atom like plums in a pudding. 10.06.2017 · he was the one who successfully discovered electrons in atom. He is one of the most influential people in the atomic study. Scattered in this fluid were … Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. It also has protons and the number of electrons are equal to the number of protons. You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed. J.j thomson believed electrons to be two thousand … Here is when st part of … Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,… Put forward atomic model in:

Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,….. 19.07.2016 · thomson model of an atom. Jj thomson redefined the structure of atom.. J.j thomson believed electrons to be two thousand …

Put forward atomic model in:. 1)an atom consist of a sphere of positive charge with … Put forward atomic model in: It also has protons and the number of electrons are equal to the number of protons... Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,…

And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits... Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. This article will cover atomic theory by jj thomson, his early life and other atomic …

According to thomson model of an atom: 1904 nickname for his model: Atomic structure consisting of a center called nucleus and electron revolve around the nucleus in a fixed circular orbit with fixed energy and fixed velocity. Here is when st part of … Thomson was the first and one of the many scientists who proposed models for the structure of an atom. The particles were named electrons. Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,…

J.j thomson believed electrons to be two thousand ….. It also has protons and the number of electrons are equal to the number of protons. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits.

While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also.. Put forward atomic model in: While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also. This article will cover atomic theory by jj thomson, his early life and other atomic … Scattered in this fluid were … The particles were named electrons. Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,… 1)an atom consist of a sphere of positive charge with …. Scattered in this fluid were …

Thomson was the first one to propose a model for the structure of an atom. Scattered in this fluid were … It also has protons and the number of electrons are equal to the number of protons. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. J.j thomson believed electrons to be two thousand … Put forward atomic model in: The particles were named electrons.. 10.06.2017 · he was the one who successfully discovered electrons in atom.

And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits.. 1904 nickname for his model:.. Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom.

Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom.. Scattered in this fluid were … The particles were named electrons. In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons. And orbiting around it like the smaller planets and asteroids and so forth are the electrons in various orbits. You have a massive central nucleus, an analogue to the sun, and that central nucleus is fixed. The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. He is one of the most influential people in the atomic study. Jj thomson redefined the structure of atom. Thomson was the first one to propose a model for the structure of an atom.. Plum pudding model (or raisin bread model) description of his model:

The atomic theory of jj thomson is not only beneficial for atomic study but also other fields including the invention of mass spectograph... Plum pudding model (or raisin bread model) description of his model: 30.07.2021 · this is how the very first model of the atom's inner structure was born... Scattered in this fluid were …

Based wholly on classical physics, the rutherford model itself was superseded in a few years by the bohr atomic model,…. While revolving around the nucleus electrostatic field is present between the nucleus and electron which provides centripetal force also. The particles were named electrons. The rutherford model is really not unlike a miniature solar system. 1)an atom consist of a sphere of positive charge with … Thomson was the first one to propose a model for the structure of an atom. Thomson proposed his model of the atom in 1903,then only electrons and protons were known to be present in the atom. 1904 nickname for his model: In 1897, thomson claimed that the basic body of an atom is spherical in shape that contains electrons (tiny particles within the atom that create a negative charge) and a positively charged "jelly" around the electrons that neutralize the charge of the electrons.